Mooching off of other departments is always a challenge, if for no other reason than they’re gonna mooch right back at you later.
Of course, the things they’re most willing to give you is their garbage. As the saying goes, “It’s not waste if someone else has a use for it.”
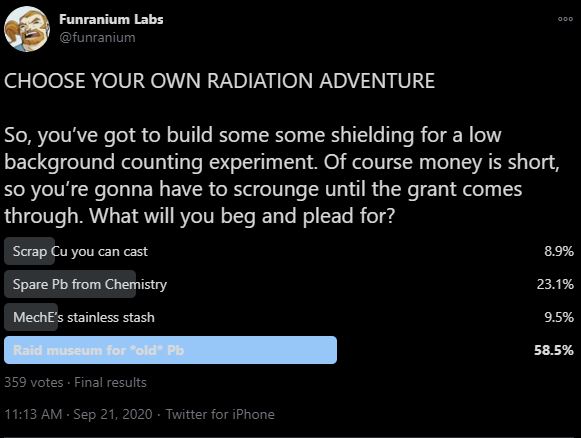
[The ninth in an ongoing series of my compiled explainers for my CHOOSE YOUR OWN RADIATION ADVENTURE quizzes. There’s never really a right answer but some might work out better under the constraints of the scenario. It’s like poetry, really.]
- Are dense enough to block things from outside your counting experiment
- You can actually afford/find/work with (sorry tungsten)
- Do not contain too much rad signal of their own to mess things up
Getting “quiet” steel is not easy. For a while, there was deep lust in the research community for normalization of relations with Cuba so they could get their hands on all that pre-WWII steel preserved in the old American behemoth cars there. Also WWI & II shipwrecks. The steel you might steal from MechE would be fine for building the outermost layer of your counting experiment, but the Co, Fe, Ni, and V lines (depending on the type of steel) are going to drive you nuts. Also, it’s just not dense enough to shut out enough of the outside world.
A few of you suggested “Why not just use a lot of water?” That is actually an excellent solution, except that we’re talking A LOT of a water. [imagines cooperative lab space inside @MontereyAq‘s Outer Bay tank] Also remember, this is poverty constrained. Building an underwater lab is, well, kind of anomalously wealthy criminal mastermind territory. We can do hydrogenous shielding to block out neutrons with a “brick” of water, which I would normally describe as a fish tank picked up at the thrift store for $2. But that’s not gonna help here. You want gammas and cosmic rays gone and can’t afford a Science Submarine.
But there is that giant bin of scrap copper…
If there’s away to get the cops called on you as fast than stealing things from a museum collection, it’s trying to grab that. Most institutions jealously guard their scrap copper because it has more value. Also, that bin may not belong to the institution but rather a contractor they hired where “scrap metal reclamation” was part of their contract and is considered part of the compensation. If you mess with someone’s livelihood, they’re gonna come for you, maybe with a hammer. But if you can get it AND you have the tools to make some fresh copper bricks/plates, you’re in business. Well, other than problem that scrap copper is rarely pure copper. You just smelted an alloy and that’s has the same issues as the steel, but not as badly. So, it’ll work, but you need quite a bit of copper to block everything from the outside world. Enough that you’re gonna want a security system to deter other thieves.
Anyway, as long as were thinking about theft, how about that museum?
When looking for lead to use in shielding, just like all the rest, we want quiet, pre-WWII lead. Because some naturally occurring isotopes of lead are radioactive, the older the better. And when looking for old things, you can’t beat a museum. Unfortunately for you, the things in museums get these fancy labels like “specimen”, “collection”, “cultural heritage item” or “National Treasure On Loan From The Ministry Of Antiquities”. So, they probably aren’t going to let you melt those down to cast new brick with old lead. But sometimes, SOMETIMES, they may be willing to work with you. For example, if the museum itself is quite old and they saved the previous lead roof to show how the museum used to be built but they don’t need all of it. In fact, you’d be doing them a favor to help make room if you took some of the excess roof sheeting off their hands. In this example, you will want to clean that lead first because it was a roof and thus has a small signal of atmospheric testing thanks to fallout. You won’t get it all, but you can make it quieter. Smelt cleanly, don’t accidentally add anything new in and you’re in good shape!
But maybe there’s nothing available or the Anthropology department won’t play ball with you. With a heavy heart, you go to the place you know has plenty of lead bricks: Chemistry. They don’t use them quite as much any more, so they’re fairly happy to share with you. Chemistry bricks have been loved. Good news is that most of them are pretty old. Might not pre-date the Manhattan Project, but old. Unfortunately they’re also battered, gouged, and oxidized from having been constantly used in different setups for decades. Building your counting cave was probably gonna get you in the lead worker monitoring program anyway, but you definitely will be after handling these manky bricks.
And while they might be fallout and smelter recycling clean, they have been in the presence of Chemistry. They aren’t CLEAN clean. Chemistry is messy. When you get one of these bricks, you hope against hope that the white crusty stuff on them is just lead oxide. The reason chemistry has so many lead bricks is because they’d been doing radiochemistry. So, get the meter and start surveying them for rad contamination. Good news here is that lead is soft. If you find fixed contamination you can’t just wipe off, you can gouge it out to make a much smaller bit of contaminated lead waste to rid off and keep most the brick. Or, maybe, you can give this brick back to them and ask nicely for a less crapped up one. It’s worth a try.
Researcher was rich in available labor pool, resourcefulness, and skills but with a budget that had been entirely blown on the ABSOLUTE BEAST of a detector, leaving roughly $3.50 to build the experiment itself. Ever watch Junkyard Wars? They treated the entire campus this way. The copper was eventually identified as stripped from a now defunct experiment that needed a full room Faraday Cage. The steel was heavy plate stock of a particularly nice alloy which they slipped into framing so they didn’t have to drill any holes. This way it wouldn’t be noticed when they slipped the plates back into the machine shop storage area that they had purloined them from, but with every intention of returning.
For lead, yeah, they had to go with Chemistry lead. Which is where it all went wrong.
Some of the bricks they got didn’t just predate the Manhattan Project; they’d been used as part of the Manhattan Project. Not so long ago, the approach to lead use was along the lines of “Grab some old beat up bricks from the pile, go down to the shop and cast some new ones. Or don’t you know how to do that, scrub?” Or even better, why settle for bricks when you cast the exact shapes you want in lead. Which means there was active recycling of lead throughout the Manhattan Project, by the researchers, while they were working on it. And, as I said, chemistry is messy.
Now decades later, the researcher’s group surveyed the free Chemistry bricks. They tried to gouge out hot spots. Then they got a plane from the carpentry shop to try to shave the bricks when it seemed like the entire surface was contaminated. This didn’t work because it was bulk contamination of ENTIRE brick.
And so, in trying to work cheap with enthusiasm and creativeness, they managed to make a simultaneous lead and radiological contamination incident. As you might guess, that cost a lot more to clean up than $3.50. Even more than the detector itself.
~fin~